A New Way to Deliver Gene Therapy Directly to Muscle
Engineered Viruses Target Muscle Cells Using Natural Fusion Proteins
Muscle diseases, such as Duchenne Muscular Dystrophy (DMD), are devastating conditions that cause progressive loss of muscle strength. In many cases, these disorders are genetic, meaning they are caused by mutations in a gene that’s necessary for healthy muscle function. For instance, in DMD, the gene that produces the protein dystrophin is defective. Without this protein, muscles become weak and damaged over time.
Gene therapy offers a promising way to treat such diseases. The idea is to deliver a working copy of the missing or faulty gene directly into the patient’s muscle cells. If successful, the muscle cells can start producing the protein they were lacking, potentially stopping or even reversing the disease’s progression.
However, delivering genes specifically to muscle cells is not as easy as it sounds. One of the biggest challenges in gene therapy is getting the therapeutic gene to the right place in the body, at the right time, and without side effects. Most of the time, viruses are used to deliver genes because of their natural ability to enter cells. But standard viruses don’t know the difference between a muscle cell and a liver cell—they’ll infect whatever they can. This lack of precision can be dangerous and limits how gene therapy can be used.
A team of scientists, led by Dr. Douglas Millay and colleagues, came up with a brilliant idea:
“What if we could engineer viruses that ONLY target muscle cells—and ignore everything else?”
In a groundbreaking study published in Cell (2023), the researchers achieved exactly that—by hijacking a process that muscle cells already use in the body: membrane fusion.
Understanding the Science: How Do Muscle Cells Fuse?
During muscle development and repair, muscle cells (called myoblasts) must fuse together to form large, multi-nucleated muscle fibers (myofibers). This fusion process is absolutely essential for building and maintaining healthy muscle tissue. Two proteins are at the center of this process:
- Myomaker
- Myomerger (also called Myomixer or Minion)
These two proteins live in the membranes of fusing muscle cells and enable them to merge, creating the long fibers that make up our muscles. They are only active during muscle growth or repair, which means healthy, resting muscle cells don’t express them.
The researchers asked:
“If these proteins are so good at fusing membranes, could we use them to help viruses fuse with muscle cells—just like muscle cells fuse with each other?”
The Experiment: Giving Viruses a Muscle Identity
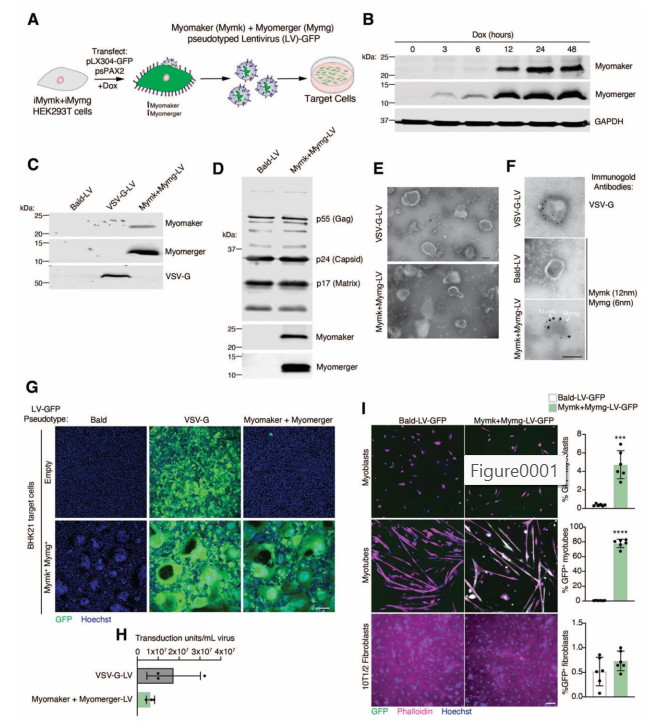
Viruses need special proteins, called fusogens, to enter cells. These fusogens act like molecular tools that allow the virus to merge its membrane with the host cell’s membrane and deliver its genetic material.
Traditionally, viruses used in gene therapy—like lentiviruses or AAVs—carry fusogens from other viruses (like the VSV-G protein from vesicular stomatitis virus). But these proteins are not specific to any one tissue. That’s why viruses using them can end up infecting many types of cells, not just muscles.
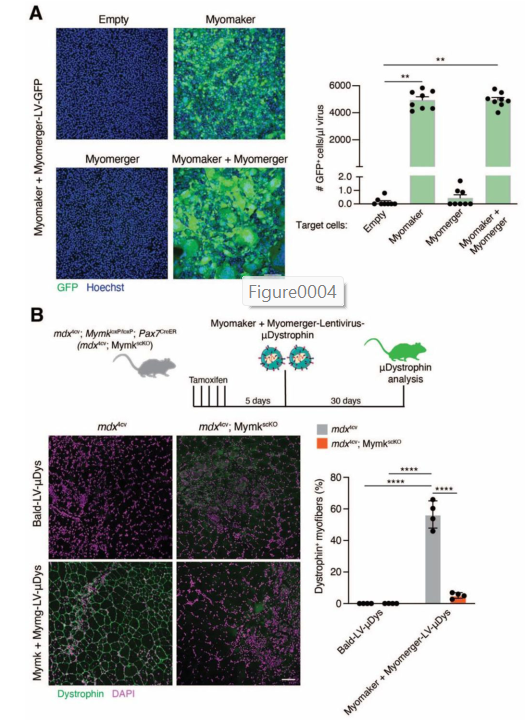
To solve this, the team replaced the viral fusogen with the muscle-specific proteins Myomaker and Myomerger. They genetically modified the viruses to express these two proteins on their outer surface. This process is called “pseudotyping”—essentially giving the virus a new identity.
Now, these engineered viruses could only fuse with cells that also express Myomaker and/or Myomerger—which, as we know, are mostly regenerating muscle cells.
This approach was tested first in lab dishes (in vitro). When exposed to different types of cells:
- The engineered viruses only entered muscle cells that were in the process of fusing.
- They did not infect skin cells, fibroblasts, or other non-muscle cells.
That was a strong early signal that the strategy could work.
Testing in Mice: A Model for Duchenne Muscular Dystrophy
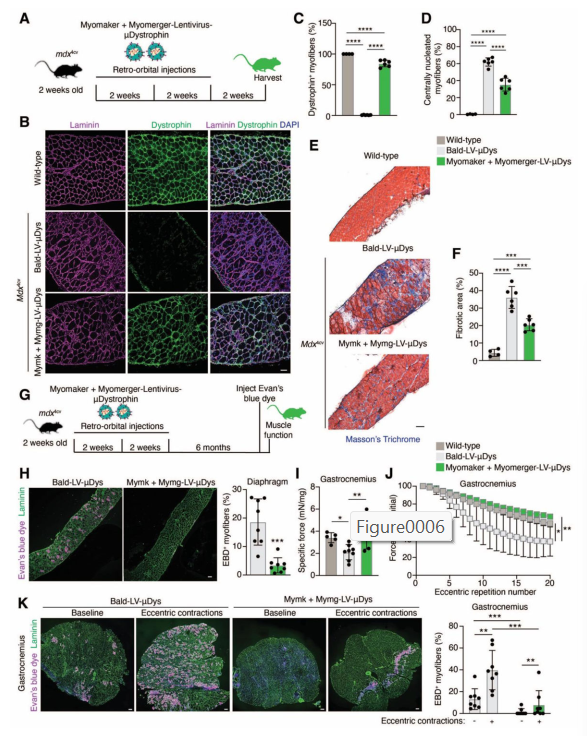
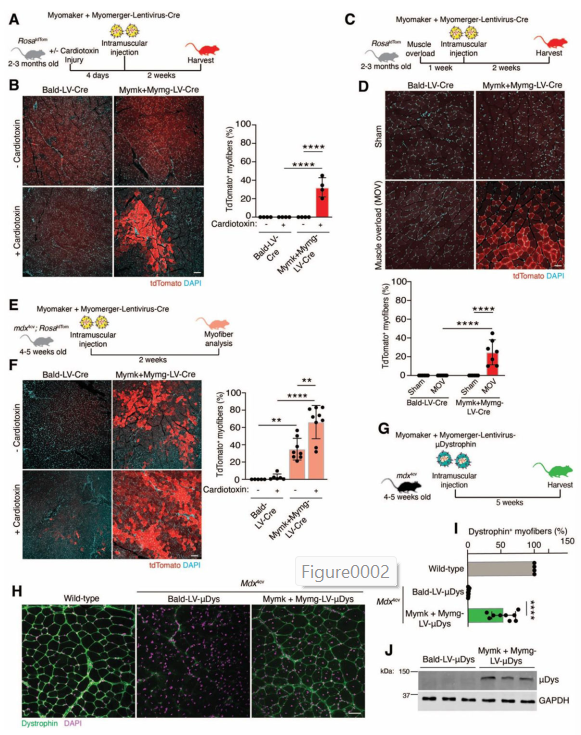
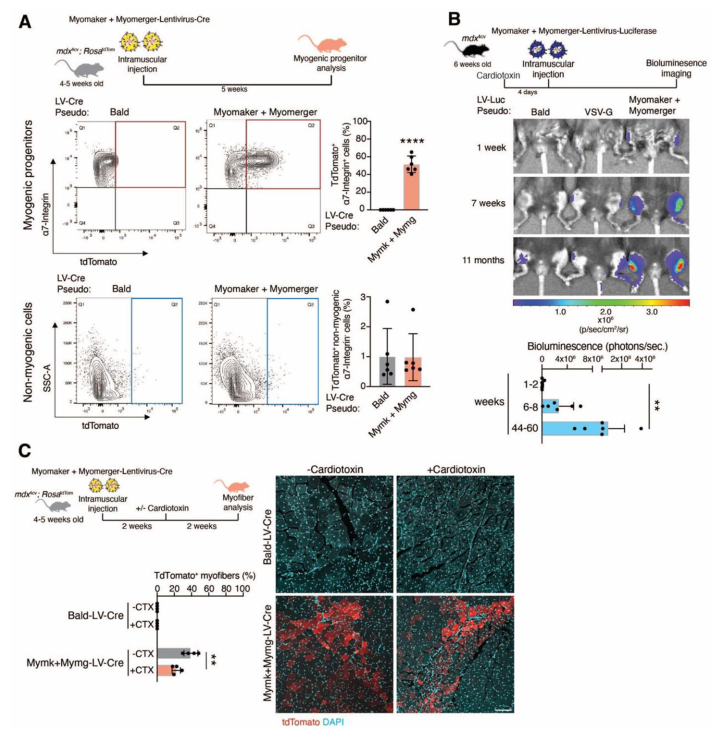
Next, the team moved on to testing their new virus in live animals. They used mdx4cv mice, which are a widely used model for Duchenne Muscular Dystrophy. These mice lack the dystrophin protein and show many of the same muscle problems as human DMD patients.
The researchers loaded their virus with a miniaturized version of the dystrophin gene (called μDys or “micro-dystrophin”) that’s small enough to fit inside a virus but still provides functional benefit.
Then they injected the engineered virus:
- Directly into individual muscles (like the tibialis anterior in the leg)
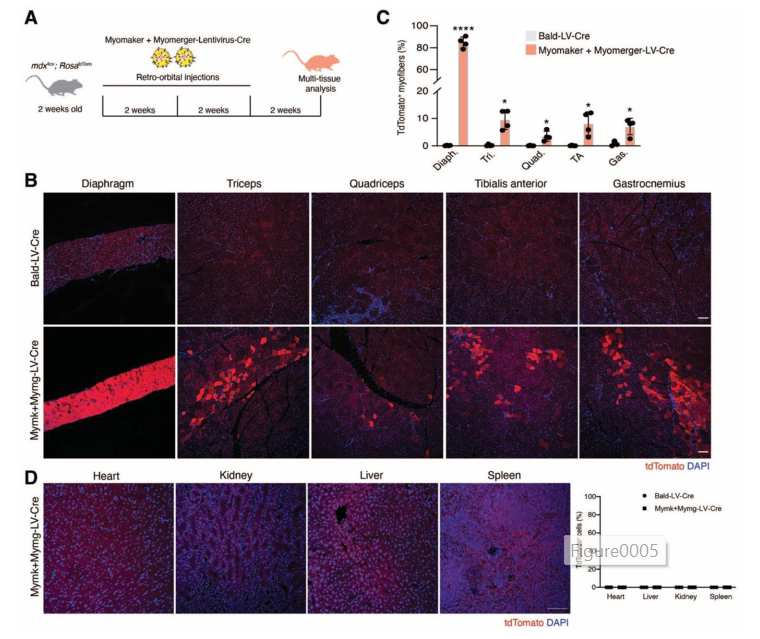
- Systemically, into the bloodstream (via retro-orbital injections)
What did they see?
- ✅ In muscles undergoing repair (due to injury or disease), the virus was able to enter the cells and deliver the gene.
- ✅ In healthy, uninjured muscle, where the Myomaker and Myomerger proteins aren’t active, the virus had little to no effect.
- ✅ Most importantly, the virus only affected muscle tissue—it didn’t enter the heart, liver, kidney, or other organs.
That level of specificity is extremely rare—and very promising for future therapies.
Real Results: Restoring Dystrophin in Muscle
After the viruses were delivered, the scientists looked at whether the muscle cells actually started producing dystrophin again.
- In local muscle injections, they saw strong dystrophin expression in many muscle fibers—while control mice showed none.
- In systemic injections, they saw up to 90% of muscle fibers in the diaphragm expressing μDys.
- The treated muscles showed less fibrosis, fewer damaged fibers, and improved strength.
They also tested whether the effect lasted over time—and it did. Even 6 months after treatment, dystrophin levels were still detectable, and muscle function remained improved.
Can the Virus Be Re-Used?
One big problem with many current gene therapy viruses (like AAV) is that you can only use them once. After the first injection, the immune system remembers the virus and will attack it if it ever comes back.
But surprisingly, the engineered virus in this study did not trigger a strong immune response. The researchers were able to give the mice multiple doses of the virus over time—and each time, it worked better.
In fact, with three doses, they reached nearly complete correction in the diaphragm muscle—something never seen before in similar studies.
Why This Strategy Is a Game Changer
Let’s break down why this discovery matters so much:
- ✅ Precision Targeting: The virus only infects regenerating muscle cells—not healthy muscle, not the liver, not the heart. That means fewer side effects.
- ✅ Gene Delivery Where It’s Needed: Diseases like DMD involve ongoing muscle damage and repair. The virus takes advantage of this by targeting the exact cells that are active and ready to fuse.
- ✅ Re-Dosable: Unlike other gene therapy methods, this one can be given more than once without being blocked by the immune system.
- ✅ Lasting Effects: The treated muscles not only showed immediate improvement—they stayed healthy over months.
- ✅ Modular Platform: This approach could be used not just for dystrophin, but for delivering any therapeutic gene to muscle tissue.
Limitations and Next Steps
Of course, there are still some questions to answer:
- Does it work in humans? This study was done in mice, and human muscle biology is more complex.
- What about heart muscle? The current strategy doesn’t target cardiac muscle, which is also affected in DMD.
- Is it safe long-term? Lentiviruses integrate their genes into the host DNA, which carries a small risk of disrupting normal genes.
But the researchers are optimistic. They suggest that this virus could be used alongside other therapies, such as AAVs for heart targeting, to create a more complete treatment.
Conclusion: A New Era in Muscle Gene Therapy
This study marks a major step forward in the field of gene therapy. By using the body’s own fusion proteins, scientists created a virus that knows exactly where to go—to the cells that need it most.
In the future, this strategy could lead to personalized, efficient, and safer treatments for many muscle-related disorders, not just DMD.
It’s a beautiful example of how understanding basic biology—in this case, how muscle cells fuse—can unlock powerful new ways to fight disease.
The future of muscle gene therapy is looking stronger than ever.








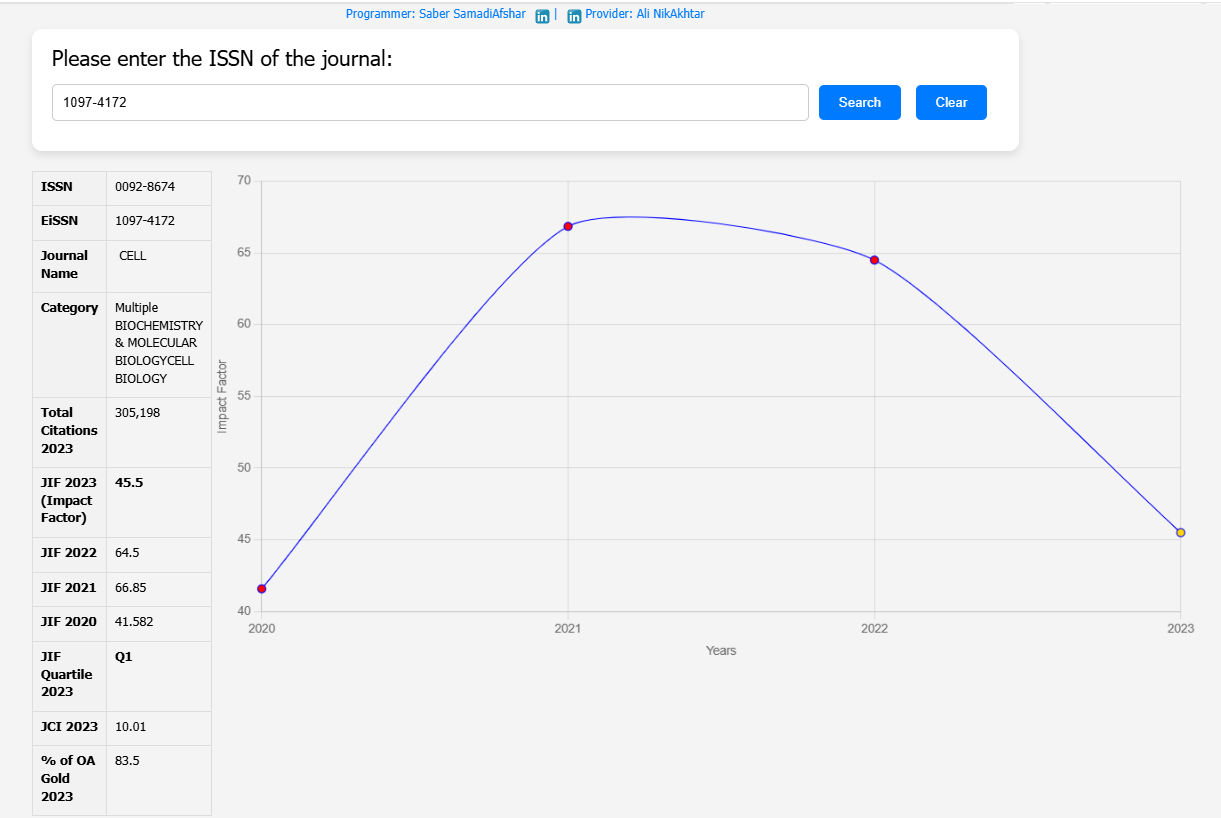
| Field | Details |
|---|---|
| Title | Enveloped viruses pseudotyped with mammalian myogenic cell fusogens target skeletal muscle for gene delivery |
| Authors | Sajedah M. Hindi, Michael J. Petrany, Elena Greenfeld, Leah C. Focke, Alyssa A. W. Cramer, Michael A. Whitt, Ramzi J. Khairallah, Christopher W. Ward, Jeffrey S. Chamberlain, Vikram Prasad, Benjamin Podbilewicz, Douglas P. Millay |
| Corresponding Author | Douglas P. Millay (douglas.millay@cchmc.org) |
| Publication Date | May 11, 2023 (final) / Available in PMC: June 17, 2024 |
| Journal | Cell |
| Keywords | Skeletal muscle, gene therapy, pseudotyped viruses, Myomaker, Myomerger, dystrophin delivery, Duchenne Muscular Dystrophy (DMD) |
| Methods Used | AAV/lentiviral pseudotyping, in vivo gene delivery, histology, RT-qPCR, western blot, functional muscle tests in mdx mice |
| Study Type | Preclinical gene delivery platform development in mouse model |
| DOI | 10.1016/j.cell.2023.03.033 |